Point To Ponder:Ī trick to remember to distinguish between a cation and an anion is to say to yourself the following:Īnion - ‘I get annoyed with negative people.’ What Type of Ion Forms When an Atom Gains Electrons? Some examples of anions are Iodide ion (I - ), chlorine (Cl - ), hydroxide (OH - ). Therefore, these elements possess a net negative charge. All non-metals gain two or more electrons and do not lose any proton. What is an Anion?Īn anion carries a negative charge that gets attracted towards the anode in electrolysis. In the above, we can see that atoms on losing electron (s) become positively charged however, the number of protons before and after the ion formation remains the same. What Type of Ion Forms When An Atom Loses Electrons?įor example, when atoms like Na, Ca, Mg, K loses electrons, it becomes positively charged by losing electrons accordingly: 
When an atom loses one or more electrons, it now has more protons, or positively charged particles, than electrons, or negatively charged particles. This is because the number of protons equals the number of electrons in an atom and the charges balance out. 
As you know that an atom is electrically neutral. But what is a cation?Ī cation is a positively charged ion that gets attracted towards the cathode in electrolysis. An electrolyte is a solution that contains ions and in drinking the sports drink, you consume cations. Have you ever gulped down a protein shake after becoming dehydrated from vigorous physical activity? Maybe a trainer, your coach, gym teacher, or even a sports drink commercial might have told you that consuming the beverage would help you replace your electrolytes. What do you find common among the battery, your heart, and fertile soil? The answer to this question is, it requires cations to work. Now, Let’s Understand what is a Cation with a Real-Life Example: Here, these ionic bonds are formed between positively charged ions called cations and negatively charged ions called anions. In Chemistry, we call ionic compounds chemical compounds that are formed of two or more ions held together by an electrostatic force of attraction these electrostatic forces form bonds called ionic bonds. Now, we will understand what is an ionic compound. What is a Charged Atom Called?Ī charged atom is called an ion and what are ionic compounds made of let’s understand this in detail: What is an Ionic Compound?įrom the above text, we understood what an ion is in Chemistry. 
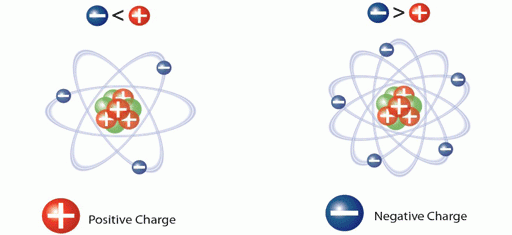
Also, the negative charge or an electron an atom holds is always equal and opposite to the positive charge or protons of an atom. It becomes a cation on losing an electron and becomes an anion when it gains an electron. What Do You Mean by Ions?ĭo you know what does ion mean and what type of ion forms when an atom loses electrons or what happens when an atom gains an electron?Īn ion is a particle that possesses an electrical charge. This page will help you understand what is an ion with its types and the ion formation. Negative ions are Cl -, O 2-, S 2-, Br -, etc. If we name the ions which are positively charged, then we have the following list: Positively charged ions are called cations, while a negative ion is known as an anion.Īn ion is an atom or a molecule that carries some charge either by losing or gaining an electron. You have been hearing since childhood that there are ions in our body, where some are positive and some negative. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
